Clinical Trials Explained – What They Are and How You Can Join
Ever wonder why new medicines appear on the market? Behind every drug is a series of clinical trials that test safety and effectiveness. Think of it as a big, organized experiment where real people help scientists figure out if a treatment works. If you’re curious or need a new option for a health issue, knowing how these studies run can open doors.
How Clinical Trials Are Structured
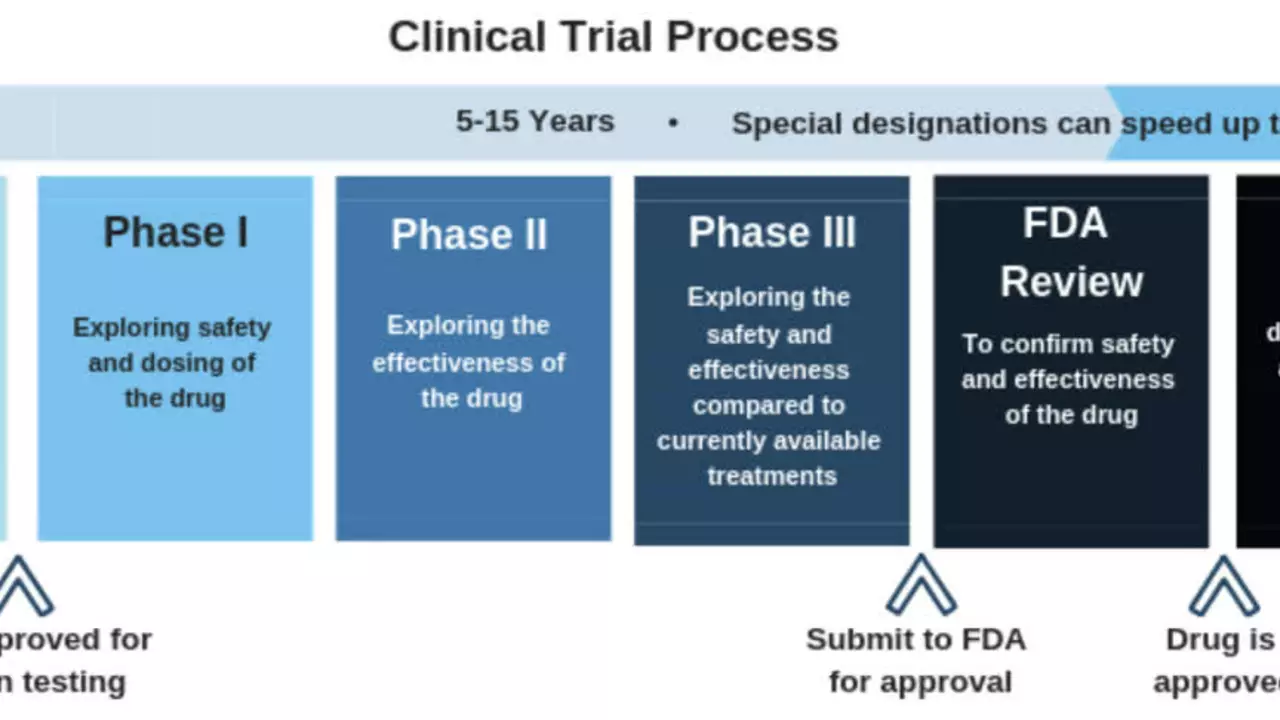
Trials aren’t random; they follow four main phases. Phase 1 is the first test on humans, usually healthy volunteers, to see if the drug is safe and to figure out dosage. Phase 2 moves to a small group of patients who actually have the condition—here researchers check if it helps while still watching safety. In Phase 3, hundreds or thousands of participants join; this is the big proof‑of‑concept that decides whether the medicine can get approval from regulators like Health Canada or the FDA. Finally, Phase 4 happens after a drug hits the market, collecting long‑term data and spotting rare side effects.
Each phase uses controls, placebos, or blinding so results stay trustworthy. A placebo looks like the real treatment but has no active ingredient, helping researchers know if improvements are due to the drug or just the hope of getting better. Blinding means participants—or even doctors—don’t know who gets what, preventing bias.
Finding a Trial That Fits Your Needs
If you think a trial could be useful, start with reputable registries. ClinicalTrials.gov is a free U.S. database that lists thousands of studies worldwide. Canada’s own site, Health‑Canada Clinical Trials Registry, works the same way. You can filter by condition, location, age, and even whether travel is required.
When you spot a study, read the eligibility criteria carefully. Some trials need specific disease stages, certain lab values, or a history of previous treatments. Others are open to anyone with the diagnosis. If you meet the basics, reach out to the trial’s contact person—usually a research coordinator—to ask about the screening process.
Before signing up, make sure the study has ethical approval from an Institutional Review Board (IRB) or Research Ethics Board (REB). This ensures your rights and safety are protected. Ask for a plain‑language consent form; it should explain what will happen, any risks, how long you’ll be involved, and whether you’ll get paid or receive free medication.
Joining a trial can feel like a big decision, but many participants say the experience gives them hope and deeper insight into their own health. Even if you don’t end up on the treatment arm, regular check‑ups and monitoring often improve overall care.
Ready to take the next step? Make a list of your current medications, recent lab results, and any questions you have about the study. Bring that to your doctor—most physicians are happy to help evaluate whether a trial is appropriate for you.
Clinical trials keep medical progress moving forward, and you can be part of that story. By understanding the phases, knowing where to look, and asking the right questions, you turn a complex process into a practical option for better health.